Core Technologies

Confocal laser scanning microscopy
Confocal laser scanning microscopy enables imaging at high optical resolution and contrast. By collecting sets of thin optical slices at different depths within a thick object the three-dimensional structure of the latter can be reconstructed. We use a Leica SP5II setup equipped with 63x oil and glycerol objectives for high resolution imaging and with blue, green, red and far red laser lines.
Fluorescence resonance energy transfer microscopy
Fluorescence or Förster resonance energy transfer (FRET) is based on the energy transfer between two light-sensitive molecules. We use this method to investigate protein-protein interactions (intermolecular FRET) as well as conformational changes within one molecule (intramolecular FRET) in real-time and in living cells or tissue slices. The respective proteins are labeled with two spectrally distinct fluorophores, one serving as the ‘donor’ transferring the excitation energy to the other called the ‘acceptor’. This energy transfer largely depends on the distance between them, which means FRET can be used as a very sensitive molecular ruler We record the fluorescence of donor and acceptor molecules using a Zeiss Axioobserver Z1 epifluorescence microscope equipped with DualView2 and Evolve camera.
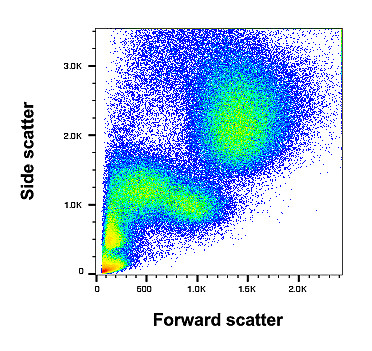
Fluorescence activated cell sorting (FACS) is a powerful tool to analyze cell populations and sort them with respect to the expression of characteristic markers. These characteristic surface proteins are labelled with antibodies that are coupled to a fluorescent dye. Based on size, granularity and fluorescent labeling, different cells can be distinguished. Our lab owns a sorter that enables us to robustly measure and separate primary cardiac cells as well as transfected cells from cell culture.
This imaging possibility provides us with non invasive in-vivo parameters of the mouse heart. The most relevant parameters for our experimental investigations are the left ventricular wall thickness (Fractional shortening, FS) and the endsystolic and –diastolic chamber diameter. Our ultrasound employs a very precise technique for generating reproducable images with a range of 0.1 mm (Vevo Imaging System der Firma VisualSonics). This are important conditions to image the mouse heart with a wall thickness of 1 mm and a heart rate of 600 bts/min.

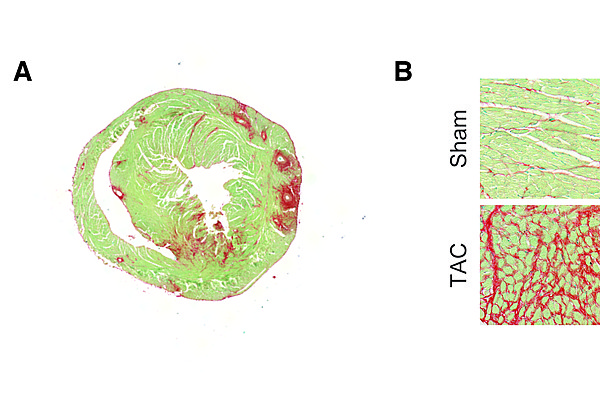
Myocardial fibrosis is assessed on a routine basis by immunohistochemistry. Paraffin-embedded or cryo-myocardial heart sections are stained for collagens (marker for fibrosis) using a Sirius Red/Fast Green staining.
In panel A, a typical 8 μm thick myocardial section is stained with Sirius Red/Fast Green. Panel B depicts representative images of control or fibrotic myocardium. The characteristic fibrosis in red is the used for quantification.
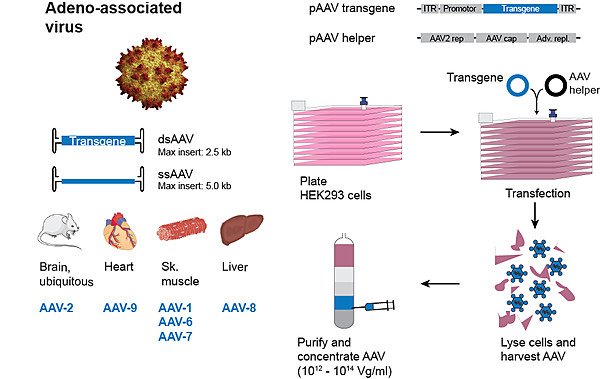
Viral vector core
Recombinant viral vectors are capable of achieving long-term and efficient transgene expression in cells and model organisms. The core facility aims to provide state-of-the-art viral vector technology to investigators for basic science research. We provide high-titer high-quality research grade adenoviruses, adeno-associated viruses (AAV) and retroviruses that can be used for both in vitro and in vivo applications. In addition, we offer consultation on strategy and choice of viral vector (serotype) and also design and production of custom vector tailored to the specific needs of the investigator. Virus stock production takes about 4-6 weeks.
Adeno-associated virus (AAV)
Adeno-associated virus is currently the most widely used viral vector for gene therapy. AAV vectors allow for persistent transgene expression and low toxicity in model organisms both in vitro and in vivo.
AAV vectors carry a payload of up to 4.5 kb and can be packaged into different serotypes (AAV1, 2, 3, 4, 5, 6, 7, 8, 9,10 and 11). All AAV preparations are purified by iodixanol gradient ultracentrifugation and are concentrated to titers ranging from 1012-1014 Vg/mL. On request, purity of AAV preparations can also be assayed for proportion of viral capsid proteins by subjecting virus preparations to polyacrylamide gel electrophoresis and silver staining
Moloney murine leukemia virus (MMLV)
MMLV, a type of retrovirus, is the most efficient vehicle for permanent gene modifications in mammalian cells. MMLV efficiently infects only dividing cells. Our lab has recently developed a MMLV-based strategy to transduce non-myocyte cells within the heart. MMLV preparations are concentrated and purified using ultracentrifugation to titers ranging from 1010-1012 Vg/mL.